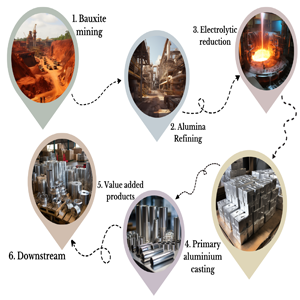
Aluminium Mining
ALUMINIUM :
Aluminium is the most profuse metallic part in the earth's crust. Due to its strong similarity to Oxygen, Aluminium does not occur in nature in its pure rudimentary state and is found in joint forms such as oxides and silicates. In order to produce the pure metal a decrease reaction must take place.
Aluminium is extraordinary for the metal's low density and for its ability to resist deterioration due to the phenomenon of passivation.Structural components made from aluminium and its alloys are vital to the aerospace industry and are important in other areas of transport and structural materials. The most useful compounds of aluminium, at least on a weight basis, are the oxides and sulfates.
Despite its occurrence in the environment, aluminium salts are not known to be used by any form of life. In keeping with its occurrence, it is well tolerate by plants and animals. Because of its occurrence, potential biological roles, helpful and otherwise, for aluminium compounds are of ongoing interest.
Aluminium Process :
Most smelting process involves direct reduction of the ore by Carbon to metal and Carbon dioxide. The process of extracting a metal from its oxide is in general referred to as smelting. Some of the subsequent steps concerned have been outlined below
Aluminium is the most profuse metallic part in the earth's crust
1. The Aluminium shaped by the electrolytic reduction of alumina dissolve in an electrolyte (bath) mainly contain cryolite (Na3AlF6) sinks to the bottom of the Cell from where it is collected and sent to a melting or holding furnace, that is used to hold the hot metal and uphold desired temperature for casting.
2 Al2O3 (dissolved) + 3C(s) ------> 4Al (l) +3CO2 (g)
2. The molten Aluminium is then mixed with desired alloys to obtain specific trait and cast into Ingots for convey to fabricating shops.
3. In the Fabrication Shops, the molten Aluminium or Aluminium alloys are re-melted and poured into casts and cooled.
4.Molten Aluminium may be additional heated to remove oxides, impurities and other active metals such as Sodium and Magnesium, before casting.
5.Chlorine may also be bubble through the Aluminium to further remove impurity.
Aluminium is theoretically 100% ecological without any loss of its natural qualities. According to the International Resource Panel's Metal Stocks in Society report, the global per capita stock of aluminium in use in society is 80kg. Much of this is in more-developed countries rather than less-developed countries. Meaningful the per capita stocks and their unsurprising lifespan are important for planning recycling.
Recovery of the metal via recycle has become an important facet of the aluminium industry. Recycling was a low-profile action until the late 1960s, when the growing use of aluminium beverage cans brought it to the public awareness.
Recycling involves melting the scrap, a process that requires only 5% of the energy used to create aluminium from ore, though an important part is lost as dross. The dross can experience a further process to extract aluminium.
MINING AND PROCESSING :
Aluminium is mined from bauxite-contained mining areas since the production of aluminium is entirely reliable on extraction from bauxite. It is not found in free form and is the most abundant metallic element in the Earth's crust. Bauxite is the only ore containing 20-25 percent of aluminium from where the metal is extracted and commercially produced. The open pit or surface mining process is carried in this case since bauxite is mostly found near surface areas where the layers of soil contain a mixed amount of various minerals, iron oxides and titanium dioxide. It is because of the iron from where bauxite gets its deep red color.
The mined bauxite is generally composed into really small pieces, loaded into trucks, railroad cars or conveyors and transported to crushing and washing plants, before being shipped to alumina refineries, which are normally located close to bauxite mines. Differently from the other metal ores, bauxite does not require complex processing and beneficiating the ore can reduce the amount of material that needs to be refined, making the process simple and getting the impurities removed simultaneously.
HOW IS ALUMINIUM OBTAINED?
Aluminium is basically obtained by using a technique called the Bayer's process developed by Karl Joseph Bayer, an Austrian chemist in 1888. Since bauxite is an aluminium ore, aluminum is thus produced by separating aluminium oxide from iron oxide present in bauxite.
This process allows bauxite to be mixed with caustic soda or sodium hydroxide and then heated under pressure after which the sodium hydroxide dissolves into aluminium oxide forming sodium aluminate whereas the iron oxide remains solid and is separated by filtration. The next step includes aluminium hydroxide to be introduced to liquid sodium aluminate resulting in aluminium oxide to precipitate or come out in solid form. These crystals are then washed and heated to form pure aluminum oxide, a fine white powder known as alumina. Alumina is a hard substance and is useful in many ways such as water purification, to make ceramics and other building materials, but mainly for extracting pure aluminium. This pure aluminium is obtained by electrolysis process where alumina is dissolved in molten cryolite.
EXTRACTING AND REFINING:
A two-step process is executed for the production of aluminium which involves refining bauxite to obtain alumina apart from removing impurities and then smelting alumina to produce aluminum. All forms of aluminum is produced from alumina which is obtained from the Bayers process using the Hall-H roult electrolytic process. Purified alumina contains 0.5 to 1 percent water, 0.3 to 0.5 percent soda, and less than 0.1 percent other oxides.
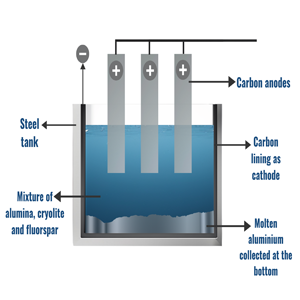
REFINING THROUGH ELECTROLYSIS :
The process of electrolysis is carried out to extract aluminium from aluminium oxide (alumina). This is performed with the help of a steel vessel coated by graphite. Since the melting point of alumina is very high, cryolite and fluorspar are added for the purpose of reducing it. This mixture is then put into an electrolytic cell which is made up of iron lined by a layer of carbon inside which serves as the cathode. A huge number of carbon rods are then dipped in the mixture where these rods act as anode.
When the electric current is made to pass through the electrolytic cell, the mixture melts and molten aluminium metal is produced at the cathode whereas on the other hand the oxygen is liberated at the anode and on reaction with the carbon rods gives carbon dioxide. This molten aluminium metal formed due to the separation of aluminium ions from alumina settles at the bottom of the vessel since it is heavier than cryolite from where it is collected and later on cooled to convert it to its solid form.
Electrolytic Reduction Of Aluminium Oxide (Alumina)
Al2O3 ------------------------> 2Al+3 + 3O-2
Aluminium oxide Aluminium ion Oxide ion
At Cathode:-
+3e
Al+3 -----------------------------> Al
Aluminium ion Aluminium Metal
At Anode:-
2e
O-2 ----------------------------> O
O + O ----------------------> O2
C(s) + O2(g) --------------------------> CO2(g)
Carbon Oxygen Carbon dioxide
TOP ALUMINIUM PRODUCING COUNTRIES :
ANNUAL ALUMINIUM USAGE:
ALUMINIUM PRODUCTION IN THE WORLD :
History has it that Guinea contains the world's largest bauxite reserves and is the biggest exporter of the ore while Australia tops the world in mine production. France was the first country of large-scale bauxite mining. In the United States, Arkansas was a major supplier of bauxite before, during and after World War II. But today, the metal is predominantly mined in countries like Australia, Africa, South America and the Caribbean with China and Russia being the topmost countries for its production following other countries like Canada, India and UAE.
(thousands of tonnes) |
||
Related Mining