Beryllium Mining
BERYLLIUM :
Beryllium is an extremely toxic metal and if bare to it, at or above the threshold values, it can lead to a chronic beryllium disease or an acute beryllium disease. Toxic contact to beryllium is most often thru an inhalation pathway. Beryllium has an assortment of effects. Some beryllium combines with a protein and is deposited in the liver, spleen and kidneys, but the beryllium when bound with a biological protein, can result in the chronic form of the disease which is supposed to be a late reaction immune response. The major toxicological effects of beryllium are on the respiratory tract, purposely the lungs and their alveoli.
Beryllium and its sole characteristic led to it being used widely in a variety of industries prior to his know toxic effects. Today it is know that beryllium is a highly toxic material which results in irresistible toxic effects on the lungs. There has been a radical increase in the rule in beryllium use so as to protect those that directly handle and work with the metal. With these system, beryllium is fairly safe to work with and use in a variety of products and industries. In the following text, there will be an account of beryllium puzzling history and toxic effects on the respiratory system of man.
Beryllium has the symbol Be. In the older chemical literature, beryllium is called glucinium after the Greek word glykys meaning sweet, because of Vauquelin's initial account and surveillance of Beryllium. Beryllium's atomic number is 4, its atomic weight is 9.01 and in its pure metal form it melts at 1278 degrees Celsius.
Following Copper's patent of the beryllium alloy, Charles II in 1921, was conspiracy by beryllium's light weight, extreme stiffness, high heat absorption and interesting nuclear cross section. Charles II was looking at beryllium and its unique characteristics to try and see if there was any possible for it to be used in any manufacturing practices. It turned out that Charles II was on to something.
Several property accounts for beryllium metal's substantial value in modern industry. It has a very high stiffness to weight ratio and low density and it has the strength of steel without steel's weight. It also has a low coefficient of thermal expansion, has an oddly high melting point and has good thermal and electrical conductive properties. Also, alloys containing beryllium are corrosive resistant and small amounts of beryllium in an alloy give this alloy a higher resistance to metal fatigue. These properties make the beryllium metal very useful in the aerospace, nuclear power, and electronics industries, along with the automobile and computers industries.
The Beryllium Corporation of America, founded by Lester Hofheimer in 1927, was the first commercially producing mining company of beryllium-containing materials. Andre w Gahagan, decided to get into the beryllium industry in 1929 and formed the Beryllium Development Corporation, and later, gained control of the Beryllium Corporation of America and distorted its name to the beryllium corporation in the year 1932. The patents and founding of several beryllium corporations sky-rocket between the years 1921, when Brush Laboratories was started to 1932 when Andre w Gahagan was trying to start a small monopoly.
Initially, in the 1930s, small amounts of Be-copper alloys, Be-oxide compounds a d tinny beryllium were made but their growth in production was enthused on the whole, through the increased demand of beryllium-copper products used in World War II. Along with the greater than before demand of World War II, a third US producer of beryllium connected products, artificial beryllium oxide mainly for their use in the bright lamp industry. Beryllium was preliminary to truly expand in its application from tinny alloys to their use in fluorescent lights.
During and right away following the second world war, a lot of research was being done in the area of nuclear fission and beryllium physical and chemical individuality, lead to beryllium being of great notice to the nuclear scientists. This work, as well as all the previous work in the metallic alloys and fluorescent light industry, was done prior to any valid knowledge of the potential hazards of beryllium. Therefore, all this work was done without the use of any protection or precautions, which came back to do some serious damage to those who worked with beryllium in a straight line and the industry.
As early as the 1930s, the period when beryllium foodstuffs were being made in a high amount, there were reports of a beryllium related disease this appeared in the European medical text. These claims/reports went on the whole unnoticed or disregarded in the United States allegedly because the findings failed to show a clear and direct relationship of beryllium causing the illness. This may be the sole reason but bearing in mind that the United States was heavily dependent on these beryllium foodstuffs in their military labors of World War II, as well as being heavily invested in the beryllium alloys, there may have been other heavily weighing factors which the administration did not want to explain so they choose to just say that the European literature was methodically unsound.
MINING AND PROCESSING:
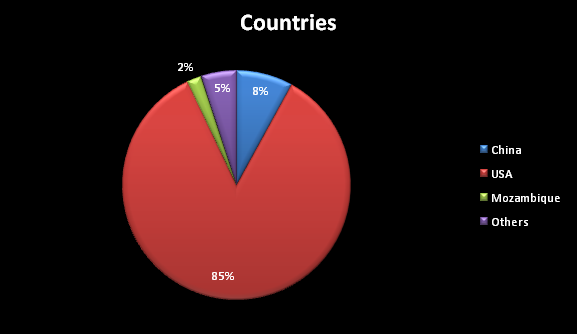
The metal beryllium is mined by its extraction from the beryl ores. It does not occur free in nature. The mining of raw minerals such as bertrandite ore and beryl ore from the Earth's crust and volcanic rocks is required. Apart from beryl, beryllium is found in the mineral bertrandite, has become a major ore of the metal in the recent years. This bertrandite mineral is found in certain volcanic rocks derived from granite. The state of Utah provides 90% of world supply from the whole of United States production in mining the bertrandite ore. Otherwise, a majority of beryllium ores are mined in China, Mozambique, Nigeria and Brazil.
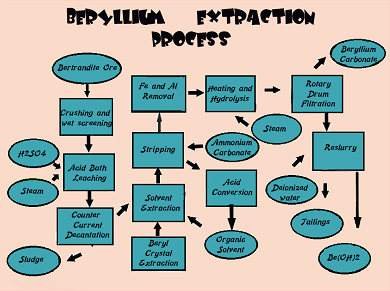
A relatively abundant metallic element in the Earth's crust, beryllium, has not been widely used due to the high cost of production and its limited consumption. Beryllium is mainly extracted in the United States, with smaller quantities coming from China, Africa, and South America. The extraction process uses leaching of the ore in sulfuric acid to produce a sulfate, which undergoes solvent extraction to remove impurity elements. The fraction containing rich amount of beryllium from solvent extraction is made to react with ammonium carbonate to yield a complex carbonate that can be separated into a pure form of beryllium hydroxide which is the basic material used for producing all useful forms of beryllium. The metallic form of beryllium is produced from the hydroxide using a magnesium reduction process to convert beryllium fluoride to beryllium pebbles and magnesium fluoride. The pebbles are further refined to >99.5% purity by vacuum melting and powdered for conversion into useful shapes by a conventional powder metallurgy pressing and consolidation process.
HOW IS BERYLLIUM OBTAINED?
Beryllium is commonly extracted from only two types ores namely beryl (Be,3Al2(SiO3)6) and bertrandite (Be4Si2O7(OH)2) ores. The beryl ore is melted in industrial furnaces, solidified and crushed, then treated with sulfuric acid to produce a water-soluble sulfate. The bertrandite ore is crushed, made into slurry and treated with sulfuric acid to form a sulfate. Sulfate solutions are required to undergo a series of chemical extraction steps to eventually produce extremely pure beryllium hydroxide. This beryllium hydroxide is considered to be the common input material for copper beryllium alloys, beryllia ceramics and pure beryllium metal manufacturing. On heating beryllium hydroxide forms the oxide, which becomes beryllium chloride when combined with carbon and chlorine. Electrolysis of molten beryllium chloride is then used to obtain the metal. The entire procedure is explained in the production of beryllium.
EXTRACTING AND REFINING :
The beryl ore seems to have more content of beryllium compared to the bertrandite ore because of which it becomes an extremely difficult process to refine the beryl ore alone. However, the refining process for both the ores is the same.
Beryl ore being harder in nature comparatively to bertrandite, is melted in an electric arc furnace and the molten material obtained is thrown into water to produce a fine powder known as frit. In the case of bertrandite, the ore is required to only be crushed unlike undergoing the heating process. This crushed ore and frit can be treated with sulfuric acid and thus the process here onwards followed is the same for both the ores. On treatment with H2SO4, the beryllium along with other metals, present in the ore get dissolved, resulting in a water soluble sulfate. This sulfate solution containing the beryllium is projected in a tank containing hydrophobic organic chemicals and hence, the solvent extraction process is executed.
The beryllium now reacts by attaching itself with the organic materials present whereas the other elements such as iron, aluminium and remaining impurities retain themselves in the water-based solution. This process can be repeated until the essential amount of beryllium content is concentrated in the solution.
When the concentrate of beryllium is obtained, the next phase is treating it with ammonium carbonate and heating it until the produced beryllium hydroxide gets precipitated. This beryllium hydroxide in its pure state is used in the making of copper beryllium alloys, beryllia ceramics and in various other applications including the manufacturing of beryllium metal.
The obtained beryllium hydroxide is dissolved in a solution of ammonium bifluoride and heated at a temperature of above 900?, producing a molten beryllium fluoride. This beryllium fluoride is then casted into molds and mixed with molten magnesium and heated in a crucible. This now separates pure beryllium from the waste material (magnesium slag). After separation, beryllium spheres of 97 percent purity are obtained. The excess magnesium undergoes further treatment and burned off in a vacuum furnace, thereby leaving beryllium of 99.99 percent purity. The beryllium spheres can be converted by isostatic pressing to beryllium powder which is useful in various beryllium alloys.
TOP BERYLLIUM PRODUCING COUNTRIES :
USES OF BERYLLIUM :
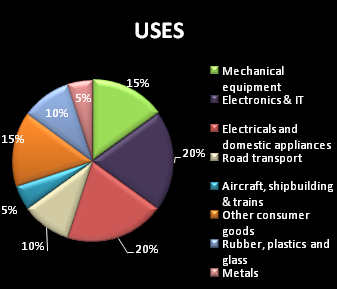
* It is useful in the aviation industry since it is used in the making of gears and cogs.
* It is used in alloys with a combination of copper or nickel for making gyroscopes, springs, electrical contacts, spot-welding electrodes and non-sparking tools.
* Used as an alloying agent, beryllium alloys are used as structural materials for high-speed aircraft, missiles, spacecraft and communication satellites.
* Beryllium is also used in consumer electronics and telecommunications and also serves in various other industrial and medical uses.
* It is also used in nuclear reactors as a reflector or moderator of neutrons.
ANNUAL BERYLLIUM USAGE :
BERYLLIUM PRODUCTION IN THE WORLD :
During the 19th century, a mixture of beryllium fluoride and sodium fluoride was used to separate beryllium by the process of electrolysis. In the beginning of the 20th century, the production of beryllium by the thermal decomposition of beryllium iodide was discovered. United States and Russia are the countries where most of the beryllium is mined and extracted whereas the state of Utah progressively contributes to two-third of world's beryllium production. Other countries known to be producing refined beryllium include China, Mozambique and Kazakhstan.
Related Mining