Copper Mining
COPPER :
Copper is a chemical part that carries the chemical symbol Cu and with an atomic number 29. It is a soft metal with glowing electrical conductivity and is somewhat flexible in its pure state and has a reddish pink gleam which is strange for metals which are usually shiny white. It discovers wide usage as a very good electrical conductor, Thermal conductor, as a building matter, and as an ingredient of a variety of copper products.
Copper is a crucial outline nutrient to every one of high plants and animals. In animals, inclusive of the humans, it is found mainly in the bloodstream, as a co-factor in a variety of enzymes, and in loads of copper based pigments. However, in enough amounts, copper can be noxious and still lethal to mortals.
Copper metal has performed a notable part in the account of humankind, which has utilized the straightforwardly reachable unrivaled metal for thousands of years in the past. A number of early civilizations have premature corroboration of utilizing copper mine. During the times of Roman Empire, copper was mainly mined on Cyprus; hence the primitive of the forename of the metal was Cyprium, which spells metal of Cyprus, later abridged to Cuprum.
Various numbers of countries, such as Chile and the United States, still possess substantial reserves of the old metal which are hauled through outsized open pit mines, nevertheless like tin there may be insufficient reserves to uphold current rates of burning up.
Copper process :
The concentration of copper in ores averages only 0.6%, and most profitable ores are sulfides, especially chalcopyrite (CuFeS2) and to a lesser extent chalcocite (Cu2S). These minerals are intense from crushed ores to the level of 10 to 15% copper by froth flotation or bioleaching. Heating this material with silica in flash smelting removes much of the iron as slag.
The procedure exploits the better ease of converting iron sulfides into its oxides, which in turn react with the silica to form the silicate slag, which floats on top of the heated mass. The resulting copper matte consisting of Cu2S is then roasted to change all sulfides into oxides.
2 Cu2S + 3 O2 ---> 2 Cu2O + 2 SO2
The cuprous oxide is converted to blister copper upon heating:
2 Cu2O ---> 4 Cu + O2
This step exploits the relatively easy reduction of copper oxides to copper metal. Natural gas is blown across the blister to remove most of the residual oxygen and electro refining is performing on the resultant material to produce pure copper.
Cu2+ + 2 e ---> Cu
Copper, like aluminium, is 100% recyclable without any loss of quality whether in a raw state or contained in an artificial product. In volume, copper is the third most recycled metal after iron and aluminium. It is predictable that 80% of the copper ever mined is still in use today. According to the International Resource Panel's Metal Stocks in Society report, the global per capita stock of Copper in use in society is 35 and 55 kg. Much of this is in more-developed countries quite than less-developed country.
The process of recycling copper follows approximately the same steps as is used to extract copper, but requires fewer steps. High purity scrap copper is melted in a furnace and then abridged and cast into billets and ingots; lower cleanliness scrap is refined by electroplating in a bath of sulfuric acid.
Copper is extracted in large amounts from the Earth’s crust by mining of sulphide ores and also oxide ores. Studies reveal about 80% of copper is produced by its extraction from sulphide ores. Chalcopyrite is a certain kind of suphide ore that can be converted to copper by a different method from silicate, carbonate or sulphate ores. Generally, the method used for the extraction of copper from its ores depends on the nature of the ore.
HOW IS COPPER OBTAINED?
The procedure of copper processing involves mining and transporting. The open-pit mining is adapted for mining copper regarding which a series of stepped benches are dug deeper and deeper into the earth over time. The machinery is then used to drill holes into the hard rock, and explosives are inserted into the drill holes to blast and break the rock to remove the ore. The resulting boulders are then ready for hauling. Most of the ores are then sent through a primary crusher, which reduces the size of the ore from boulder to golf ball-sized rocks after which it becomes easy for the extraction process to take place. Copper can be obtained by processing its oxide ores or sulfide ores:
PROCESSING OF OXIDE ORE :
Oxide ores are generally processed using hydrometallurgy. Liquid solutions are used in these processes to extract and purify copper from copper oxide ores at ordinary temperatures, in three major steps: 1) heap leaching -to leach out metals, 2) solvent extraction- to separate the liquids, and 3) electrowinning- which is a type of electrolysis helps in producing about 99.99% pure copper.
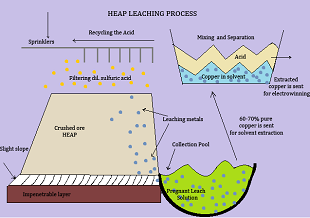
1. Heap Leaching :
The first step is the Heap Leaching process of allowing filtering chemical solutions to leach out metals. It is very commonly used for low-grade ore, which is not economical enough to be sent through a milling process. Once the mining and transporting is done, the ore is crushed to a consistent gravel or golf ball-size, the crushed ore is piled into a heap on top of an impenetrable layer, on a slight slope. The leaching reagent- dilute sulphuric acid is sprayed through sprinklers on top of the heap pile and allowed to trickle down through the heap dissolving the copper from the ore. The outcome is a “pregnant” leach solution of sulfuric acid and copper sulfate collected in a small pool. The copper compound is known to be present in concentrations of 60-70%.
2. Solvent Extraction :
The solvent extraction process allows two immiscible liquids to be stirred and separated making the copper to move from one liquid to the other. The pregnant leach solution is strongly mixed with a solvent causing the copper to migrate from the leach solution into the solvent. The two liquids then separate based on solubility, with copper remaining in the solvent solution and impurities left in the leach solution.
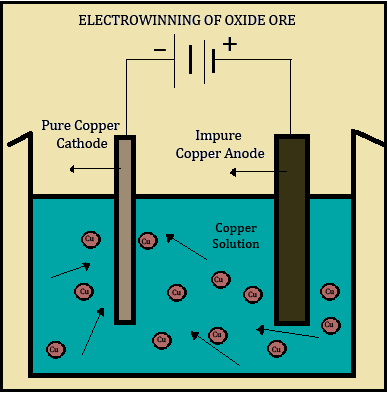
3. Electrowinning :
The final step is electrowinning. In this process, an electrical current is passed through an inert anode (positive electrode) and through the copper solution obtained previously, which acts as an electrolyte. The cations come out of the solution after which they are plated onto a cathode (negative electrode) as 99.99% pure copper.
PROCESSING? OF SULPHIDE ORE:
The sulfide ores are usually processed using the method of pyrometallurgy ?a process of extraction and purification of metals which involves the application of heat in four major steps: 1) froth flotation ?to separate the copper minerals from the gangue, 2) thickening stage results in a combination of 30% copper and other metals, 3) smelting stage further produces some impurities and a combination of copper with other metals, and 4) electrolysis gives way for finished copper cathodes which can be made into wires, plates, tubes, and other copper products.
1. Froth Flotation :
Once the process of mining, transporting, and crushing the ore to a golf-sized ball is accomplished, it is further processed and reduced to pebbles and then to sand like particles. It is then added with liquid to make it a slurry which is a combination of valuable copper ore minerals and “worthless” rock, called gangue. The froth floatation process is executed to separate the copper minerals from the gangue. Collectors which are chemical reagents are added to the slurry which bind to the copper particles, making them waterproof. With the usage of pipes air is blown into the solution to create bubbles rising up along with the copper sulfide particles as froth of copper-rich bubbles. This froth is then skimmed off whereas the gangue at the bottom is disposed off.
2. Thickening :
The froth which is skimmed is poured into large tanks called thickeners. In the thickening process, the bubbles break and solids from the froth solution settle at the bottom of the tank. Filtration is carried to remove excess water from the solids and the final product of this stage is a mixture of 30% copper and other metals and this copper concentrate is then sent to the smelter.
3. Smelting :
The copper concentrate obtained in the previous step is sent through the smelting furnace and heated up to 2,300 ?F and converted into molten liquid which is is poured into a slag-settling furnace. A combination of matte including a mixture of copper, sulfur and iron, along with other metals impurities is produced. The copper matte generated by the smelting furnace contains 58-60% copper. The molten matte is taken to another furnace called a converter to have the remaining iron and sulfur burned off. This product is referred to as blister copper, which contains 98% copper, and taken to the anode smelter. The yellow color of the blister copper turns bluish green when the oxygen in the copper is burned off in the anode smelter. The resultant is molten anode copper which is poured into molds called anode-casting wheels. The cooled anode slabs are 99% pure copper, are found to be copper-colored, have two handles molded on top, and are two inches thick, three feet wide, three-and-a-half feet high, and weigh 750 pounds.
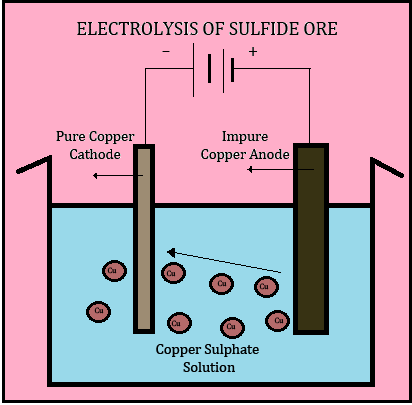
4. Electrolysis :
The final step of electrolysis refines pure copper from the sulfide ores .The copper anode slabs are hung in a large tank of copper sulfate and sulfuric acid solution which acts as the electrolyte. The cathodes are thin sheets of pure copper hung in between the anodes. The electric current is passed, and the cations leave the anode and move through the electrolyte solution to be plated on the cathode whereas the other metals and impurities also leave the anode and settle at the bottom of the electrolytic solution. The finished copper cathodes contain 99.99% pure copper and can be made into wires, plates, tubes, and other copper products.
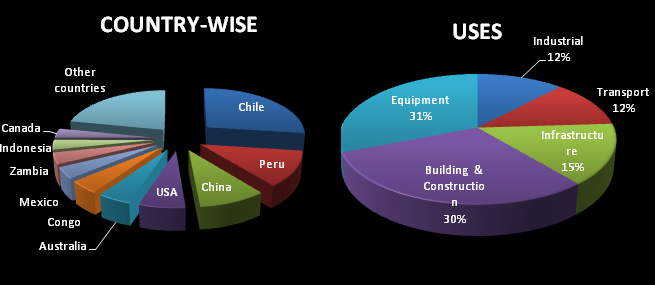
ANNUAL COPPER USAGE:
COPPER PRODUCTION IN THE WORLD :
Countries like Chile, Peru and China are the three topmost countries known for the highest production of copper across the world. USA is the fourth largest producer of copper in the world since the largest copper mine is found in Utah. Other major copper mines are found in Australia, Arizona, Michigan, New Mexico and Montana.
Related Mining