Magnesium Mining
MAGNESIUM :
Magnesium derives its name from magnesite, a magnesium carbonate mineral, and this stone in turn is told to owe its name to magnesite dumps found in Magnesia, a region in the antique Greek region of Thessaly. The British chemist Humphry Davy is said to have manufactured a mixture of magnesium in 1808 by electrolyzing damp magnesium sulfate, utilizing mercury as a cathode.
The 1st victorious industrialized manufacture was begun in Germany in 1886 by Aluminum und Magnesiumfabrik Hemelingen, depending on the electrolysis of molten carnallite. Hemelingen later became constituent of the industrial compound IG Farbenindustrie, which, for the duration of the 1920's and 30s, urbanized a process for manufacturing huge quantities of molten and fundamentally water-free magnesium chloride (now branded as the IG Farben process) as well as the expertise for electrolyzing this merchandise to magnesium metal and chlorine.
MINING AND CONCENTRATING :
Both dolomite and magnesite are hauled out and concerted by conservative methods. Carnallite is hauled out as ore or alienated from other salt amalgams that are brought to the exterior by solution mining. Naturally happening magnesium including brines are determined in large ponds by solar desertion.
EXTRACTION AND REFINING :
A physically powerful chemical reagent, magnesium structures steady compounds and responds with oxygen and chlorine in equally the liquid and gaseous state. This means that taking out of the metal from unrefined materials is a force exhaustive procedure requiring nicely tuned technologies. Commercial construction pursues 2 completely dissimilar methods: electrolysis of magnesium chloride or thermal lessening of magnesium oxide. Where power charges are stumpy, electrolysis is the cheaper means and, undeniably, it accounts for something like 75 percent of world magnesium production.
1. ELECTROLYSIS :
The process consists of two steps; firstly to perform an industrial process involving magnesium chloride and then decompose it into the magnesium metal and chlorine gas in electrolytic cells. Generally in industrial processes, the feedstocks contain different molten salts which consist of anhydrous magnesium chloride, partly dehydrated magnesium chloride or anhydrous carnallite. In order to remove impurities from the carnallite ores, by controlled crystallization from heated magnesium and potassium containing solutions, dehydrated artificial carnallite is produced.
The Dow process is performed to obtain partly dehydrated magnesium chloride. The process requires seawater to be mixed with lightly burnt dolomite in a flocculator causing precipitates of insoluble magnesium hydroxide to settle at the bottom of the tank. This Mg(OH)2 is pumped out as a slurry and then filtered and converted to magnesium chloride by treating it with Hydrochloric (HCl) acid and dried. The latter phase of smelting process indicates the final dehydration.
The anhydrous magnesium chloride is produced in two consecutive methods; either through dehydration of magnesium chloride brines or chlorination of magnesium oxide. The IG Farben process is executed for obtaining fully dehydrated magnesium chloride. On obtaining precipitated Mg(OH)2 from the Dow's process, the compound is filtered and calcined to magnesium oxide which is mixed with charcoal and formed into globules on addition with magnesium chloride solution. After it is dried, the globules are charged and heated in a furnace by carbon electrodes at a temperature of 1000-1200?C and chlorine gas is introduced through portholes in the furnace which reacts with magnesium oxide to produce molten magnesium chloride (MgCl2). This molten MgCl2 is tapped off and sent to the electrolytic cells.
The dehydration of magnesium brines is obtained by removing impurities through precipitation and filtering in the Norsk Hydro process. The purified brine obtained contains about 8.5 percent magnesium which is concentrated by evaporation to 14 percent and converted to fine particles and on further drying to water free particles, it is sent to the electrolytic cells.
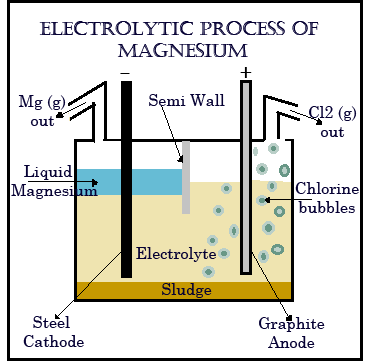
The electrolytic cells are vessels equipped with multiple steel cathodes and graphite anodes, mounted vertically and submerged in a molten salt electrolyte composed of alkaline chlorides to which magnesium chloride produced in any one of the above ways is added in concentrations of 6-18 percent. The Magnesium chloride decomposes to magnesium metal which floats to the top from where it is collected and chlorine and other gases are generated at the graphite anodes. The reaction goes like this :
MgCl2 ------------ > Mg (liquid) + Cl2 (gas)
2. THERMAL REDUCTION :
The most expensive, yet the alternate method for obtaining magnesium is through thermal reduction methodology. In this method, the dolomite is calcined to magnesium oxide and lime and reduced by silicon producing magnesium gas and a slag of dicalcium silicate. The reaction which takes place is as follows:
2CaO + 2MgO + Si -------------- > 2Mg + Ca2SiO4
The reaction being endothermic, requires necessary amount of heat to applied to initiate and sustain it. After the extraction of magnesium by the above processes, the unprocessed magnesium metal is transported to cast shops for removal of impurities, addition of alloying elements, and transformation into ingots, billets, and slabs.
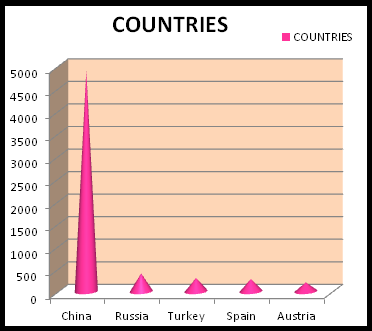
TOP MAGNESIUM PRODUCING COUNTRIES :
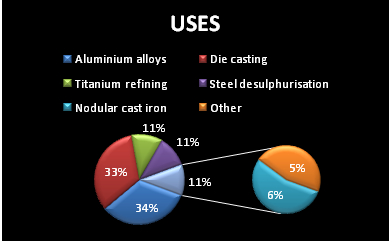
ANNUAL MAGNESIUM USAGE :
MAGNESIUM PRODUCTION IN THE WORLD :
Although magnesium bearing ores are found all over the world, the biggest producer of magnesium is known to be China. Magnesium is mined in China following other countries like North Korea, Russia, Canada, Austria and USA. China being rich in magnesite content is the reason why it continues to be the extremely huge producer of magnesium in the world, leaving the other countries behind. Also, magnesium-bearing brines are estimated to constitute a resource in the billions of tons, and magnesium can be recovered from seawater at many places along the world's coastlines. Highly concentrated magnesium reserves are found to be only in three countries- Russia, China and Korea. The United States also contribute to magnesium reserves as the Dead Sea and the Great Salt Lake in Utah are abundant in magnesium resources. The topmost countries ranking accordingly in first five positions are as follows: